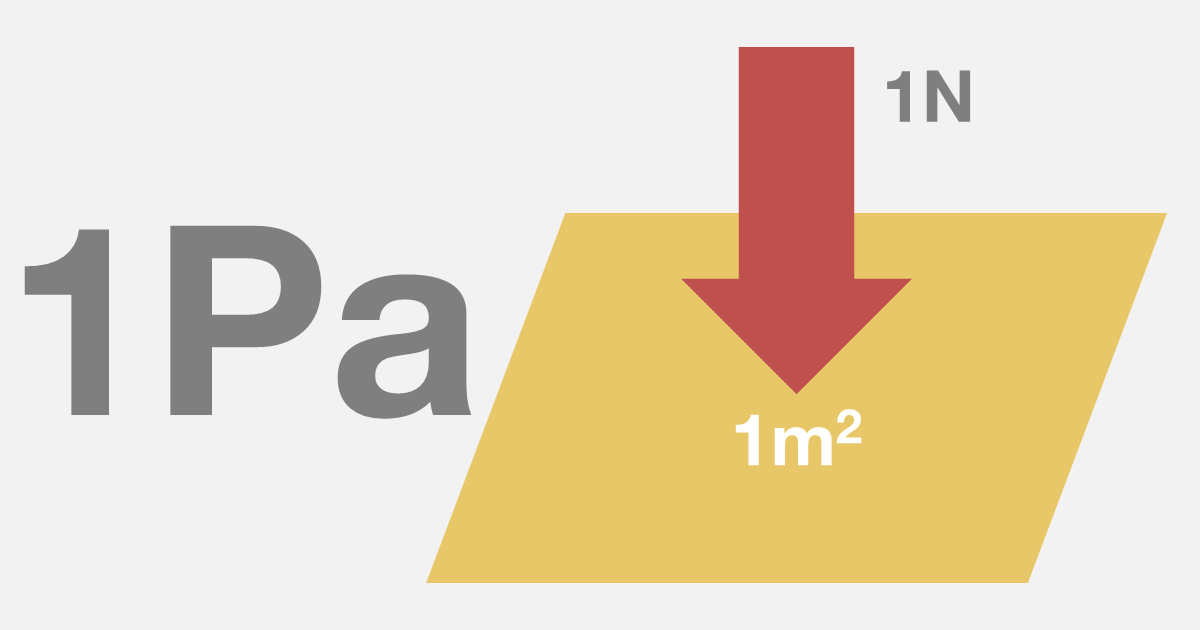
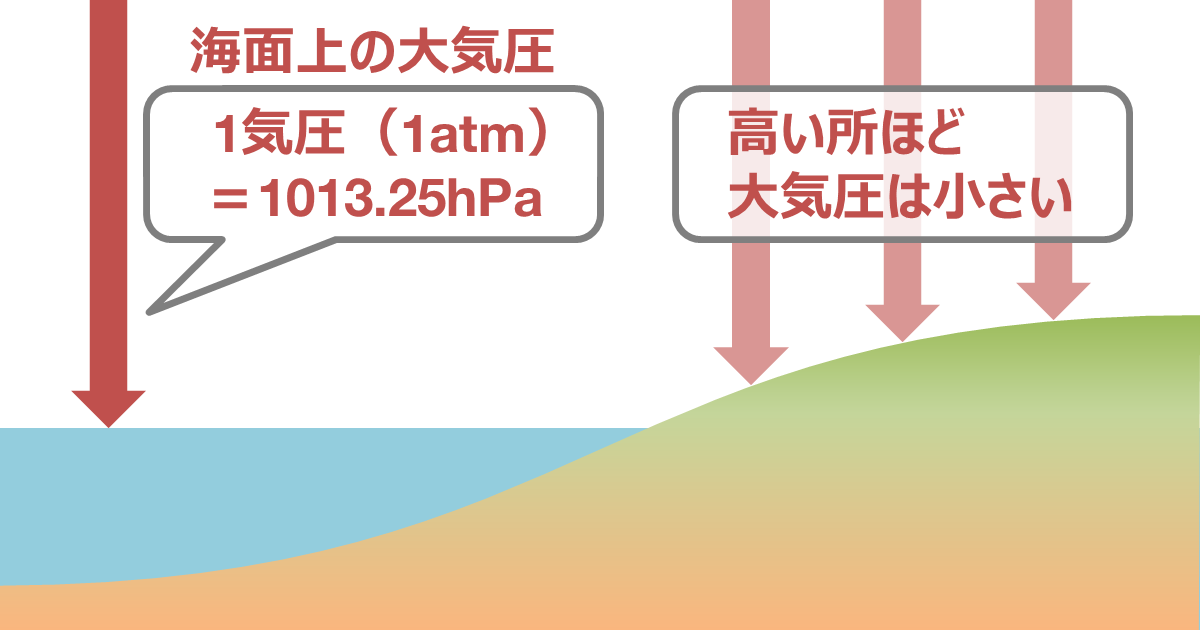
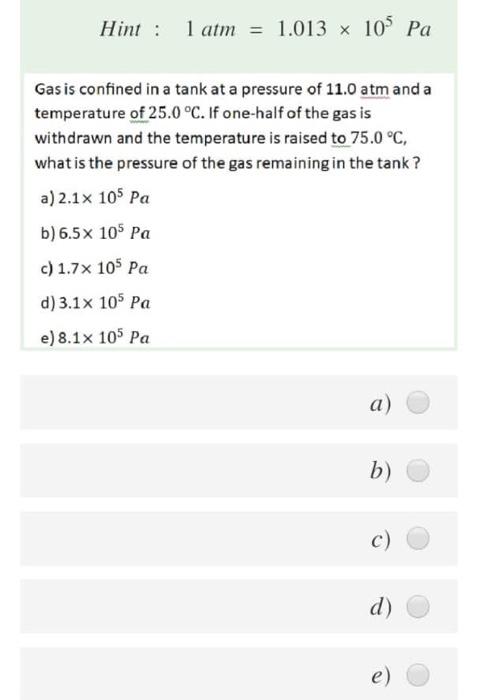
How to convert Atmospheric pressure to Pascal (atm-Pa) and Pascal to Atmospheric pressure (Pa-atm). - YouTube
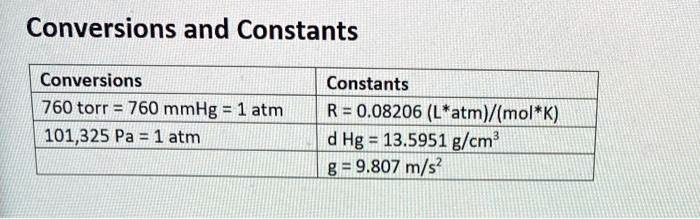
SOLVED: Conversions and Constants Conversions 760 torr = 760 mmHg = 1 atm 101,325 Pa 1atm Constants R = 0.08206 (L*atm)(mol*K) d Hg 13.5951 g/cm? 82.9.807 m/s?

Gauge pressure Most pressure gauges compare to Atmospheric (i.e. this room is at 0 Gauge) Absolute p is 1 atm more P = P gauge + 1 atm 1 atm = x. - ppt download

SOLVED: Equalities 1 atm 760 torr (exact) 1 atm 760 mmHg 1 atm 29.92 inHg 1 atm 101325 Pa 1 atm 14.7 psi Equations 9PF TF = Tc + 328F 5PC 5*C
![SOLVED: 1atm = 760. mm Hg 760.torr 1 atm 101325 Pa 101.3]=1Latm 1 nm 1x10-9 m 0.08212 M ) h = 6.63x10-34J c = 3.00 x108 m Rn = 2.18 xlO-18 J - SOLVED: 1atm = 760. mm Hg 760.torr 1 atm 101325 Pa 101.3]=1Latm 1 nm 1x10-9 m 0.08212 M ) h = 6.63x10-34J c = 3.00 x108 m Rn = 2.18 xlO-18 J -](https://cdn.numerade.com/ask_images/8a6c5e9b80cf49b1b8b4573ce740e6f2.jpg)
SOLVED: 1atm = 760. mm Hg 760.torr 1 atm 101325 Pa 101.3]=1Latm 1 nm 1x10-9 m 0.08212 M ) h = 6.63x10-34J c = 3.00 x108 m Rn = 2.18 xlO-18 J -

SOLVED: Convert 661,000 pascal, pa into torr: 1atm = 760 torr = 760 mmHg = 101,325 pa 101.325 kpa 1.01325 barr 14.696 p.s.i. 29.921 inHg (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)
The guage pressure exerted below a column of water, open to the earth's atmosphere at depth of 10 m is (density of water = 1000 kg/m3, g = 10 m/s2 and 1 atm pressure = 105 Pa)