JEE ADVANCED 2017 SOLUTION - Among H2, He2+, Li2, Be2, B2, C2, N2, O2−, and F2, the number of - YouTube

SOLVED: According to Molecular Orbital Theory, two separate Is orbitals interact to form what = orbitalls)? Molecuig A. nand n" B. 0 only C.nonly D. o and 0" E. 0 and m

06L121111H 13-19 Volkswagen Golf MKVII,5G1,BQ1,BE1,BE2 Hatchback 2.0 GTI Water Pump Thermostat Housing Assembly Generic - Mad Hornets

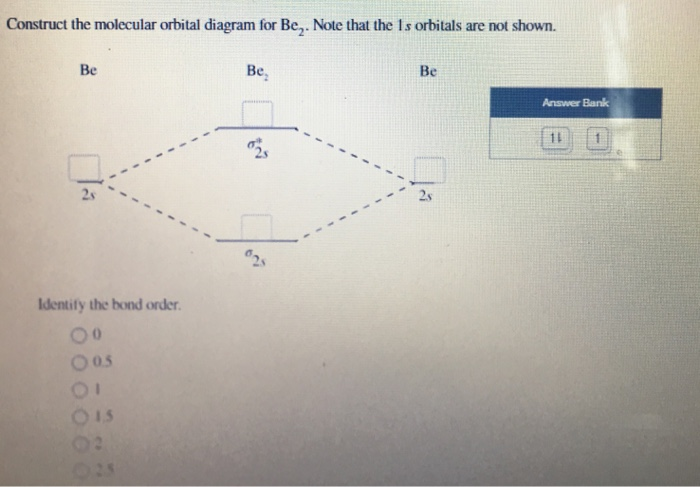
Use the appropriate molecular orbital energy diagram to write the electron configuration of the given molecule and ions. Be2, Be2+, Be2- | Homework.Study.com

Use MO diagrams and the bond order from them to answer each of the following questions. (a) Is O2 stable or unstable? (b) Is Be2+ diamagnetic or paramagnetic? | Homework.Study.com

The beautiful WW1 RAF BE2 made of wood and Irish linen - Picture of NZ Warbirds Association, Papakura - Tripadvisor